You may have heard of such abstruse biological terms as "phosphorylation" and "protein kinases", but few know that they are the most sophisticated "signal regulators" in cells. Everything, from basic cellular processes like growth and metabolism to the occurrence and progression of cancer, is under their control.
Today, we will use plain language to understand the synergetic regulatory mechanism of phosphorylation and protein kinases, and uncover the story of these two "core players" in cells.
First, the Basics: What are Phosphorylation and Protein Kinases?
Simply put, phosphorylation is like flipping a switch for proteins. By adding or removing a "phosphate group" (the equivalent of a switch button), proteins in cells can rapidly switch between an "active" or "inactive" state, thereby regulating a variety of cellular functions.
And the molecules responsible for flipping this switch are protein kinases. They act as the "dedicated electricians" inside cells, specialized in adding phosphate groups to proteins to initiate signal transduction. Corresponding to them are "phosphatases", which "turn off the switch" by removing phosphate groups to terminate signals. With this on-off cycle, cellular processes such as growth, metabolism, and division are precisely regulated.
More crucially, a single protein kinase can regulate multiple proteins, and a single protein can be phosphorylated by multiple kinases. This complex "cross-regulation" forms an extensive signal network within cells, much like an elaborate "circuit network", ensuring the orderly operation of all cellular activities.
New Research Findings: The "Hidden Mechanisms" of Phosphorylation and Kinases
In the past, our understanding of phosphorylation was largely limited to the model of "a single kinase regulating a single protein". However, the reality is far more complex.
1. Multilevel Regulation by "Signal Integrators": The Bidirectional Crosstalk Between Metabolism and Signaling
Studies by Humphrey et al. have indicated that there are numerous "signal integrators" in cells (e.g., AS160, the PFK2 family, TSC, etc.), which can be phosphorylated by multiple distinct kinases. This allows them to integrate different signals and adapt to various cellular microenvironments.
For example, AS160 is a key protein regulating glucose transport. It can be phosphorylated both by Akt kinase (activated by insulin) and AMPK kinase (activated by exercise). This explains why both insulin and exercise can promote cellular glucose uptake: essentially, two different signaling pathways achieve functional synergy by phosphorylating the same "integrator".
More interestingly, metabolism and signaling engage in bidirectional crosstalk: kinases not only regulate metabolism, but metabolic products can in turn modulate kinase activity. Take AMPK kinase as an example—it can sense the energy level in cells. When cellular energy is insufficient, AMPK is activated and initiates an "energy-saving mode" by phosphorylating downstream proteins (shutting down energy-consuming biosynthetic processes and activating energy-generating catabolic metabolism), thus sustaining cell survival.
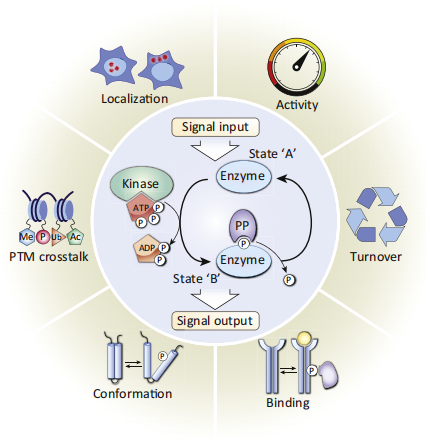
Reversible protein phosphorylation acts as a molecular switch mechanism [Humphrey, [et.al](et.al)].
2. Deciphering the "Dark Phosphoproteome": Identifying Potential Cancer Targets
Jiang et al. focused on a critical question: cells contain a large number of unannotated phosphorylation sites (referred to as the "dark phosphoproteome"). We know neither the kinases that regulate these sites nor their biological functions—and this is exactly the blind spot in cancer research.
The research team used machine learning to construct a Phosphorylation Co-regulation Network (CoPheeMap), and combined it with a kinase-substrate prediction tool (CoPheeKSA), successfully "illuminating" these "dark sites". Their key findings include:
Many of these "dark phosphorylation sites" are actually critical cancer-associated sites, regulated by certain understudied kinases—these kinases may serve as novel therapeutic targets for cancer (e.g., CDK12, previously overlooked, is now found to be abnormally activated in multiple cancers, and its inhibitors can suppress cancer cell proliferation).
This network enables the rapid identification of kinase-substrate pairing relationships. For instance, it predicted that PDK1 kinase can phosphorylate the Thr497 site of the PRKCA protein—a finding validated by experiments, providing a clear direction for subsequent research.
Why These Studies Matter: They Relate to Everyone’s Health
Dysregulation of phosphorylation and protein kinases is one of the core causes of cancer development. The unlimited proliferation and metastasis of many cancer cells stem from the dysfunction of kinase switches, leading to the disorder of cellular signal networks. For example, abnormal activation of specific kinases (e.g., the CDK family, AKT kinase) is observed in common cancers such as lung cancer, breast cancer, and liver cancer.
a) They reveal novel regulatory mechanisms of phosphorylation, helping us better understand how the dysregulation of cellular signals leads to cancer;
b) They identify more potential therapeutic targets for cancer (especially those neglected "minor kinases"), providing ideas for the development of new targeted drugs;
c) They offer more efficient research tools (e.g., CoPheeMap), enabling scientists to quickly decipher the "dark phosphoproteome" and accelerate the pace of cancer research.
Small Phosphate Groups, Big Biological Roles
Though phosphorylation is a subtle molecular modification, it governs the entire life cycle of cells—from growth and proliferation to senescence and death. As key "regulators", protein kinases are essential for maintaining cellular order. In the future, precise regulation of these "kinase switches" may provide more robust support for the precision treatment of diseases and breakthroughs in basic scientific research.
References:
Jiang, W., Jaehnig, E.J., Liao, Y. et al. Deciphering the dark cancer phosphoproteome using machine-learned co-regulation of phosphosites. Nat Commun 16, 2766 (2025). doi:10.1038/s41467-025-57993-2
Humphrey SJ, James DE, Mann M. Protein Phosphorylation: A Major Switch Mechanism for Metabolic Regulation. Trends Endocrinol Metab. 2015;26(12):676-687. doi:10.1016/j.tem.2015.09.013
