As spring begins and new journeys unfold, scientific research moves toward new horizons.
When performing ELISA experiments, have you ever faced this confusion: why do identical procedures sometimes produce precise and highly reproducible data, yet at other times result in false positives, weak signals, or distorted standard curves?
In fact, the success of an ELISA experiment lies within every key component. Coating antibodies/antigens, detection antibodies, substrates, washing buffers—these may appear to be simple consumables, but each carries a critical mission. Their mechanisms of action directly determine the specificity, sensitivity, and even the final outcome of the assay.
Today, we will take a deep dive into the working mechanisms of the key ELISA components (using the double-antibody sandwich method as an example). Only by understanding their “division of labor” can we eliminate experimental errors at the source, ensure every dataset stands up to scrutiny, and move ahead efficiently on the path of scientific research.
Core Component ①: Coating Antibody — The “Foundation” of ELISA, Building a Solid Detection Platform
The coating antibody is the first line of defense in an ELISA experiment and the cornerstone of the entire detection system. It acts as an “exclusive capture platform” for the target molecule.
Mechanism:The coating antibody is immobilized in the microwells of the ELISA plate. Through the specific antigen–antibody interaction, the target molecule in the sample is precisely anchored in the well and not washed away during subsequent washing steps, laying the foundation for signal amplification and detection.
Key impact:The coating concentration, purity, and activity directly determine the capture efficiency. Excessive coating leads to non-specific binding (high background), while insufficient coating results in poor capture (low signal). Low activity of the coating material reduces binding capacity and leads to poor reproducibility.
Core Component ②: Biotinylated Antibody–Enzyme-Labeled Streptavidin — The “Signal Amplification Team”
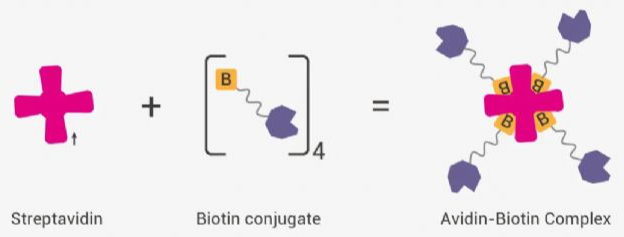
If the coating antibody is the “capturing hand,” then the biotinylated antibody–enzyme-labeled streptavidin system is the cooperative “signal amplification team.” Together they exponentially amplify the antigen–antibody binding signal and convert it into a detectable and quantifiable visual/instrumental signal, especially suitable for low-abundance targets.
Mechanism:The biotinylated antibody specifically binds to the target molecule already captured by the coating antibody, forming a primary complex (“coating–target–biotinylated antibody”), effectively labeling the target.
Streptavidin acts as the central “bridge,” with extremely high affinity for biotin (over 1000 times stronger than antigen–antibody affinity). One streptavidin molecule can bind four biotin molecules, creating a structural framework for signal amplification.
HRP is covalently linked to streptavidin and then binds specifically to the biotinylated antibody, forming a multilevel complex (“coating–target–biotin antibody–enzyme-labeled streptavidin”). After substrate addition, the enzyme catalyzes the chromogenic reaction. One target molecule can recruit multiple enzyme molecules, achieving exponential signal amplification.
Key impact:The specificity and labeling efficiency of the biotinylated antibody and the purity of streptavidin directly affect amplification efficiency. Low labeling efficiency or insufficient purity results in weak amplification, high background, and inaccurate quantification of low-abundance targets.
Core Component ③: Substrate — The “Signal Presenter” That Visualizes Data
While the biotin–streptavidin system amplifies the signal, the substrate presents it. It is the key bridge between the enzymatic reaction and data readout.
Mechanism:The HRP enzyme acts specifically on the substrate, catalyzing a chromogenic reaction. For example, TMB changes from colorless to blue (and turns yellow after stopping). The color intensity is positively correlated with the target concentration. The absorbance is then read by a microplate reader to calculate the target concentration.
Key impact:The purity, stability, and reaction specificity of the substrate affect color clarity and consistency. Low purity causes background coloration, and poor stability leads to uncontrolled reactions and inaccurate results.
Core Component ④: Washing Buffer — The “Cleaner” That Eliminates Interference
Although simple in appearance, the washing buffer is essential for removing interference and ensuring accurate detection. Each wash clears obstacles for precise measurement.
Mechanism:Its core function is to remove unbound sample impurities, unbound biotinylated antibodies, and enzyme complexes, leaving only the specific complex (“coating–target–biotin antibody–enzyme-labeled streptavidin”) in the wells, thus preventing interference.
Key impact:The number and duration of washing steps directly affect background and reproducibility. Insufficient washing leads to high background, while excessive washing may remove specifically bound complexes, resulting in weak signals.
A New Year Message for Researchers
Every set of precise ELISA data relies on the seamless cooperation of these core components. Understanding their mechanisms allows you to avoid experimental errors at the source, reduce detours, and obtain reliable results efficiently.
Reed Biotech ELISA kits provide pre-coated microplates, saving customers from the time-consuming coating process. They employ high-affinity monoclonal antibodies and optimized coating buffers for precise and stable binding; high-purity biotin and streptavidin for efficient signal amplification; substrates tested for purity and stability with strong specificity and uniform color development; and a stable washing buffer system that effectively removes unbound substances.
In the Year of the Horse 2026, may every researcher ride forward with determination, cultivate deeply in the field of science, and achieve continuous and immediate success in research outcomes.
